Week in Washington is brought to you by Michael Cohen, PhD. Tune in each week to read the latest on healthcare policy and get a glimpse of what’s on the horizon.
Week in Washington
1/14/2020
Incoming Administration
There is expected to be a flurry of potential legislation and regulations in the initial months of the new Biden Administration. At the top of the priority list is another COVID bill with an aim at improving vaccine distribution and providing more economic stimulus. While the details are still not entirely clear and will evolve once Congress begins debating the issuer in earnest but at a basic level the Biden proposal would be a nearly $2 trillion relief plan that includes funding for combating COVID (vaccine distribution, testing, etc.), economic stimulus (checks, unemployment insurance, etc.) and increased subsidies for health coverage (new Exchange benchmark for subsidies would be a gold plan, premiums capped at 8.5% for those above 400% FPL, subsidies for COBRA coverage, and funding for states and local government)
COVID
The pandemic continues to in the United States as daily cases, hospitalizations, and deaths are at the peak. Over 25,000 died this week from COVID. Complicating the situation is the introduction of more contagious COVID variants, which could worsen the situation in the coming weeks and months.
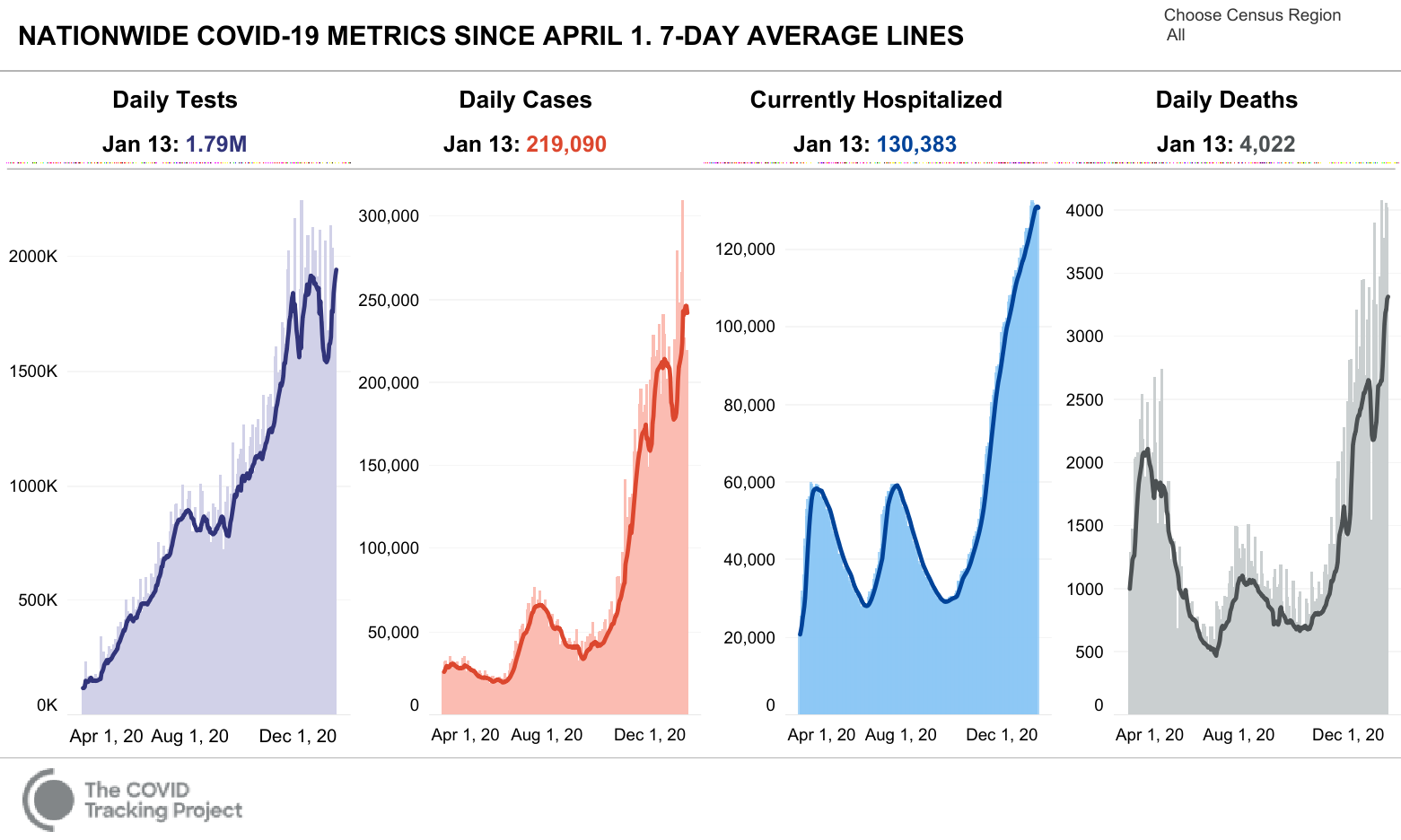
COVID Vaccine Administration
As of writing this blog, over 11 million US residents have received the initial vaccine, which is equal to about 3% of the population. However, currently, the Federal government is allocating only about 4 million additional doses a week. As Larry Levitt notes, as context, there are about 53 million people over 65, meaning it may be a while before the general populace has access to the vaccine. Johnson and Johnson’s is expected to release interim results on its’ vaccine at the end of January.
Regulations: Coming and Going
Medicare
- OMB has finished its review of the “The Contract Year 2022 Policy and Technical Change” to the MA Program rule and is expected to be published soon.
- The Pharmaceutical Care Management Association sued the Trump Administration to block the drug rebate rule from going into effect.
- The proposed $200 drug discount card for some Medicare Part D enrollees will not happen.
Medicaid
- CMS approved Tennessee’s Medicaid waiver that would change the program into effectively a block grant. The waiver approval is expected to be challenged in court and by the incoming Biden Administration.
ACA
- CMS released its final Open Enrollment numbers for the Exchanges that HHS operates. Overall 8.3 million people selected plans using the Healthcare.gov platform. This represents the first year over year increase since 2016. As several State-based-Exchanges still have Open Enrollments ongoing, the final tallies are not expected until March.
- HHS finalized part of the 2021 Notice of Benefit and Payment Parameters. While some issues like direct enrollment and 1332 waivers were included in the rule other issues like risk adjustment will be tackled in future regulations.
Previous editions:
1/08/2020: Week in Washington: The Year Ahead
12/17/2020: Week in Washington
12/10/2020: Week in Washington
12/03/2020: Week in Washington
11/19/2020: Week in Washington
11/12/2020: Week in Washington
11/05/2020: Week in Washington
10/29/2020: Week in Washington