Week in Washington is brought to you by Michael Cohen, PhD. Tune in each week to read the latest on healthcare policy and get a glimpse of what’s on the horizon.
Week in Washington
01/28/2021
Biden Executive Order
Today, President Biden released several executive orders as a way of signaling the new direction the Administration would take on the ACA and Medicaid. Executive orders are orders to create regulations and policies; in other words, on their own, they do not have the force of law. Furthermore, implementing the vision could take months, if not years, so it’s best to think of them as coming attractions. Nonetheless, you can read the Executive Order here, which includes a list of policy priorities that be found here
Here are a few highlights:
ACA
- HHS will open HealthCare.gov for a “Special Enrollment Period” from February 15, 2021 – May 15, 2021.
- While this order applies to the state Exchanges that HHS operates, several State-Based Marketplaces (SBMs) (Colorado, Washington, New Jersey, and California.) have already announced that they would mirror the HHS policy. Other SBMs could join as well.
- The Special Enrollment Period would be open to both new enrollees and current enrollees.
- KFF recently released research that 14.9 million uninsured are be Marketplace eligible.
- Prior research has shown that enrollment among low-income individuals may be higher (and therefore risk selection) when open enrollment periods align with the tax-filing season.
- Plan to make changes to regulations that:
- Affect 1332 waivers;
- “Undermine the Health Insurance Marketplace or other markets for health insurance” (e.g., short-term duration plans, association health-plans, etc.)
- Reduce affordability of coverage or financial assistance, including for dependents
Medicaid
- HHS will make a concerted effort to end regulations and policies that allow for Trump-era 1115 waivers (i.e., work requirements.)
- Change regulations to increase enrollment in Medicaid.
Biden Legislative
Reports circulated that Democrats in Congress could begin work on a reconciliation bill as early as next week.
COVID
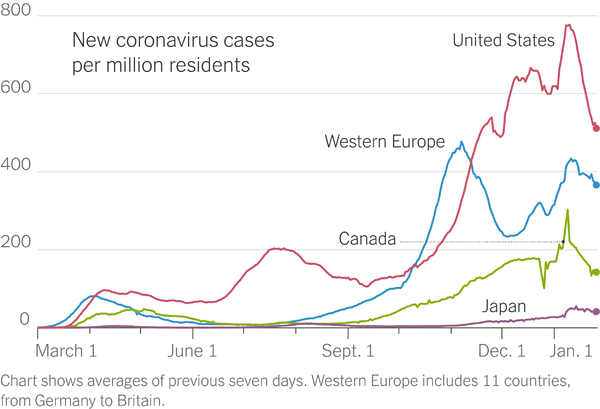
COVID cases have declined 35% over the past three weeks. Hospitalizations have also declined substantially. While deaths remain high (for example, over 4,000 people died in the US due to COVID yesterday), deaths are also expected to decline in the coming days and weeks (COVID deaths typically lag case numbers by 3-4 weeks).
The declines have been attributed to a combination of improved mask-wearing/social distance adherence, and some levels of herd immunity being reached (an estimated 100 million Americans have had COVID so far, and about 24 million people have received a vaccine shot) may be reducing the spread.
While about 25 million US residents have been diagnosed with COVID, the CDC is estimating that about 1 in 4.6 infections are being reported (and about 1 in 2 COVID hospitalizations are being reported.)
COVID Variant
While the news is good for now, there are increasing worries that the new, more contagious variants, could result in large increases in the not too distant future. For example, Paul Rumer’s modeling suggests that the combination of the new COVID variants coupled with lockdown fatigue could result in more than 300,000 additional deaths in the coming months.
Vaccines
Good
- 21 million people have received one more both doses of the vaccine (4 million who have been fully vaccinated). Last week, the US hit a hit of 1.2 million doses per day.
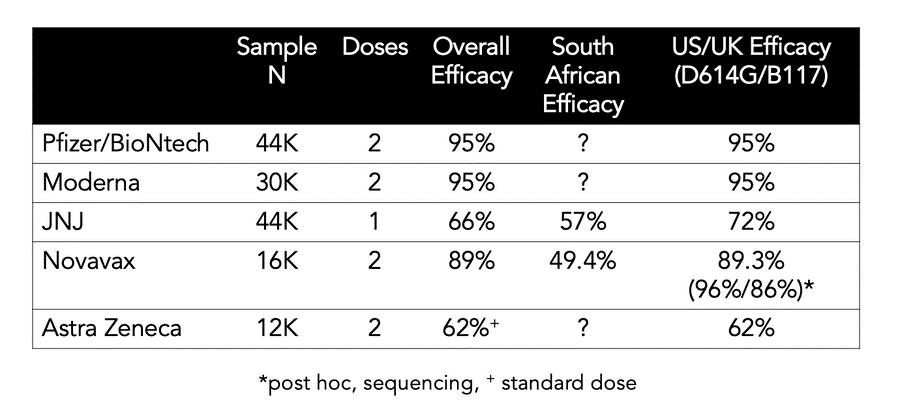
- Johnson and Johnson released results on their Phase III testing. Overall they found to have an efficacy of 66% (note that J&J vaccine regiment is one shot) although less so against the South African variant. J&J is expected to file an emergency authorization next week. Based on prior experiences, the FDA is expected to authorize in 2-3 weeks. If it’s approved the vaccine would be available to the public in March or April (there have been manufacturing delays)
- Another vaccine candidate made by Novavax reported that its vaccine is 90% effective. There is no timeline for Novavax to submit to the FDA (the data was from non-US trials). Below is a handy chart comparing the vaccines (only Pfizer and Moderna are currently approved for use in the US)
Bad
- While 1.2 million may seem a lot, as Drew Altman pointed out, in order to vaccinate 70% of the population by July 4, you’d need 2.4 million doses. To reach 70% by Labor Day, you would need 1.9 million doses. The current pace of 1.2 would mean that you’d reach 70% vaccinated around January 1. While these numbers don’t include those that have immunity through natural infection, they do highlight the challenges facing vaccine distribution.
- While the Novavax vaccine had a 90% efficacy generally it had slightly under 50% efficacy in its South African trials. J&J also had a drop in efficacy against the South African variant. All vaccine producers are currently working on boosters against this variant, highlighting the potential that COVID vaccines may need to be updated or include an additional booster in the near future.
Other HHS Announcements
- HHS announced that the Public Health Emergency would likely stay in effect through 2021
- The Biden Administration halted the Trump Administrations' plan to let more physicians prescribe opioid-treatment drugs. The Biden Administration noted that legal concerns were one of the main hurdles.
The Economy
The Bureau of Economic Analysis (BEA) released the latest US GDP figures today. Overall, the BEA found that the US economy shrank by 3.5% last year, the worst year for the US economy since demobilization following World War II in 1946. While the expectation is that the economy will grow in 2021 and US interventions reduced income losses, the economy continues to have problems, as over 18 million Americans are currently receiving unemployment benefits.
Research you can Use
HCCI conducted analysis of the difference between the Medicare Physician fee schedule and the payments received by physicians. They generally found that FFS payments were lower than PFS rates.
Previous editions:
1/21/2021: Week in Washington
1/14/2021: Week in Washington
1/08/2021: Week in Washington: The Year Ahead
12/17/2020: Week in Washington
12/10/2020: Week in Washington
12/03/2020: Week in Washington
11/19/2020: Week in Washington
11/12/2020: Week in Washington